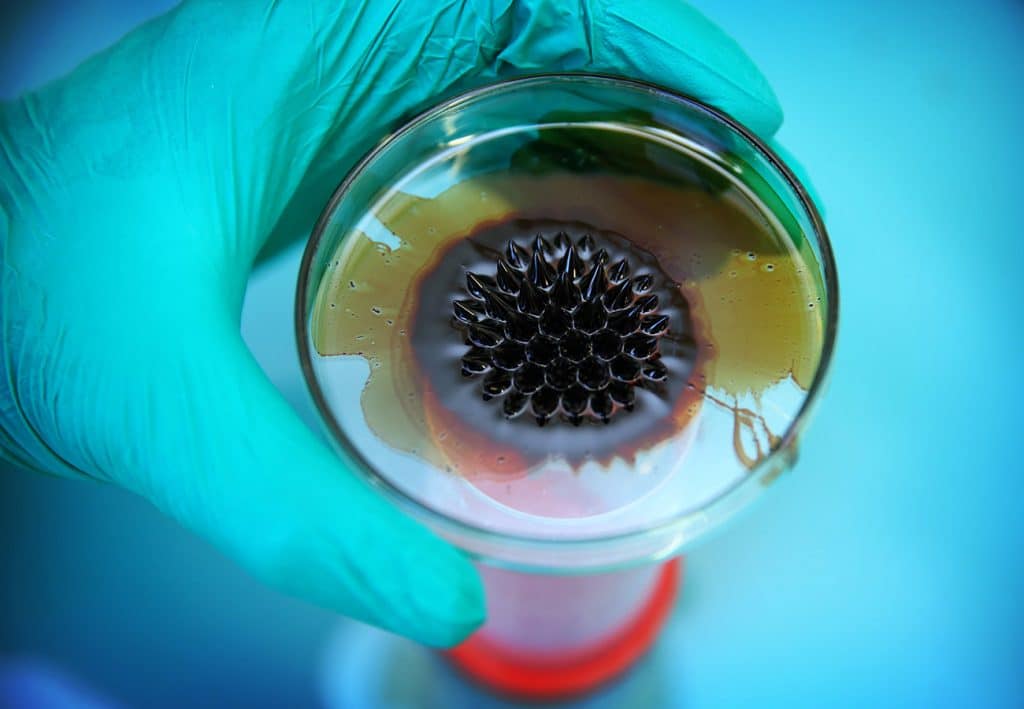
Reactivity of Nanometric phases
The physical and chemical properties of natural and engineered nanophases are different from those of mineral phases of the same chemical composition but larger in size. These properties, in particular an increased reactivity specific to their nanoparticulate state, imply new behaviours in the environment in which the team is interested (e.g. transfer in porous media, stabilisation of organic compounds in soils, adsorption of pollutants).
From the ion to the particle :
dynamics at the interfaces
Structural modifications of small nanoparticles can modify their solubility or dissolution kinetics. Dynamic processes involve exchanges between dissolved species and core atoms via atomic diffusion phenomena. The boundary between crystalline and amorphous nano-phases and polymetallic structures becomes blurred.
We seek to link the stability and reactivity of nanoparticles with their structural properties. We focus our efforts on particles of nanometric sizes (from which new properties appear <10-20 nm) up to atomic clusters of 3 to 10 atoms with properties that are still little explored.
One of the objectives is to characterise the chemical stability (dissolution) and properties (optical, electrical, catalytic) of these clusters associated with different mineral surfaces and in confined environments.
Colloid and metal transfers
The reaction mechanisms at the nanometric scale are understood in order to better understand the fate of nutrients and contaminants in natural (surface water, soil, sediments, etc.) or anthropogenic (water treatment process) aqueous compartments.
Mechanistic and holistic approaches are implemented, combining laboratory experiments, field experiments and modelling, which allow the necessary scale and complexity of transfers to be addressed. In particular, the problem of the transfer of pollutants via surface water, soils or banks, from the source of discharge (product life cycle) to the water resource (water uses) is a priority object of study. The role of colloids in natural attenuation processes is studied and remediation solutions inspired by nature are envisaged in a wastewater recycling logic.
Organo-mineral interactions
Organo-mineral interactions control OM dynamics in natural surface systems, particularly in soils. The team is interested in the role of nanominerals in the stabilisation of organic compounds with respect to microbial degradation and, conversely, in the inhibition of the growth of secondary minerals by these organic compounds.
Molecular-scale interactions and properties of organo-mineral nano-compounds are studied in order to understand the factors controlling the destabilisation of organo-mineral complexes. The approach combines work on analogues and samples extracted from natural soils. Adapted techniques for separations, quantification of OM and their dynamics (13C, 14C) and structural characterisation of organo-minerals at the nano-scale (nanoSIMS, SEM and TEM, nanoCT, synchrotrons) are implemented.
Ongoing projects
- 2022-2025: ANR SURFMEMO project. Mineral Surfaces: Origin and archive of biological information.
- 2022-2025 : National Programme ANSES 2021-EST-126 UV-FILTER. Emission, fate and environmental effects of UV filters from sunscreens - Benefits and risks of chemical or nanoparticulate substances.
- 2022-2023 : Fed. Project Rech. ECCOREV/Institute ITEM - AGREDYN. Role of aggregate structure in contaminant adsorption/desorption dynamics.
- 2020-2023 : National Carnot Water and Environment Programme NEREIDE. Nature-based Solutions for Wastewater Treatment and Reuse for Sustainable Irrigation.